It has often been said that chemistry was, and to an extent may still be, the most information-intensive of the sciences; see, for example, the article by Lyn Robinson and myself on chemical information literacy. This status is now challenged by molecular biology, with its ‘Central Dogma’ stating that information flows from DNA to RNA to proteins, and its reliance on an array of informatics systems, as noted by Divan and Royds’ recent short introduction. However, particularly to someone like myself who studied chemistry, it is interesting to reflect on the extent to which information representation and communication has gone hand-in-hand with the development of concepts and theories in chemistry, so that it is difficult to tell where the one ends and the other begins.
 I was particularly struck by this, when looking through William Brock’s short introduction to the history of chemistry. Although this is stated to be about how chemical explanations were found, and how chemical phenomena were discovered, studied, and exploited, the book is, to a remarkable extent, also a history of information representation and communication over many years.
I was particularly struck by this, when looking through William Brock’s short introduction to the history of chemistry. Although this is stated to be about how chemical explanations were found, and how chemical phenomena were discovered, studied, and exploited, the book is, to a remarkable extent, also a history of information representation and communication over many years.
Listed below are just some of the examples described by Brock.
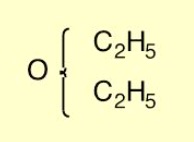
Graphical notations to represent the structures of organic compounds were developed by the Scottish chemists Couper and Crum Brown in the 1850s and 1860s, greatly assisting analysis of chemical phenomena by giving the organising principles of structure and bonding, and providing essentially the form in which organic structures are represented today in chemical information systems. 


For inorganic chemistry, the equivalent organising principle was Mendeleev’s periodic table of the elements. It is interesting that, as Brock notes, Mendeleev was led to construct his table by his search for a better way to organise the material of a chemistry textbook; the table has been used for information organisation and retrieval ever since.




Chemistry has always been a science involving a mass of observational and experimental facts and data, such that the renowned physicist Ernest Rutherford could dismiss it as ‘stamp collecting’, somewhat unfairly in view of the organising principles already developed at that time, and much further developed later. It is interesting to reflect that both the main aspects of the subject, data compilation, and organising concepts and principles, have been reflected in the information conventions and systems of chemistry.
The naming of chemical substances has always been an issue, even in the days when only a relatively small number were known (in 2015 the number of distinct substances recognised by the Chemical Abstracts Registry System reached 100 million). Brock draws attention to the significance of a conference held in Geneva in 1892, at which a group of chemists and editors of chemical journals agreed to use structural theory as the basis for giving names to substances, this form of information representation greatly improving the ability to find chemical information in handbooks and indexes. It led to what is today known as the IUPAC nomenclature. This, however, came at the cost of public understanding of the science, as the new substance names were lengthy and difficult to understand; for example, ‘citric acid’ in the new nomenclature became ‘2-hydroxypropane-1,2,3-tricarboxylic acid’. Hence the continuation of ‘common names’ for substances familiar to the general public.
Brock’s book, a condensation and also an updating of his much more extensive 1992 History of Chemistry text, is certainly a reliable and readable introduction to the history of the subject. But it also gives a clear insight as to the extent to which information representation, and information communication, both reflect and determine a subject domain.
References
Bawden, D. (2015), Storing the wisdom: chemical concepts and chemoinformatics, Informatics, 2(4), 50-67, available at http://www.mdpi.com/2227-9709/2/4/50
Bawden, D. and Robinson, L. (2017), “An intensity around information”: the changing face of chemical information literacy, Journal of Information Science, 43(1), 17-24, open access version at http://openaccess.city.ac.uk/12707/
Brock, W.H. (2016), The history of chemistry – a very short introduction, Oxford, Oxford University Press
Brock, W.H. (1992), The Fontana history of chemistry (2 vols.), London: Fontana
Chemical Abstracts Service (2015), CAS assigns the 100 millionth CAS Registry Number to a substance designed to treat myeloid leukemia, [media release] available at http://www.cas.org/news/media-releases/100-millionth-substance
Divan, A. and Royds, J.A. (2016), Molecular biology – a very short introduction, Oxford: Oxford University Press
Hepler-Smith, E. (2015), “Just as the structural formula does”: names, diagrams, and the structure of organic chemistry at the 1892 Geneva Nomenclature Congress, Ambix, 62(1), 1-28
Rzepa, H. (2012), The first ever curly arrows, [blog post] available at http://www.ch.imperial.ac.uk/rzepa/blog/?p=7234
Wikipedia (2017), IUPAC nomenclature of organic chemistry, available at https://en.wikipedia.org/wiki/IUPAC_nomenclature_of_organic_chemistry
